
COVID-19 is an acute respiratory illness borne from infection with SARS-CoV-2 – a positive single-stranded RNA virus. The SARS-CoV-2 virus shares structural similarities to SARS and Middle East Respiratory Syndrome coronavirus (MERS-Cov-1), which cause acute severe respiratory infections. The incubation period for the development of clinical COVID-19 symptoms from the time of initial SARS-CoV-2 viral infection is highly variable, ranging from 2 to 14 days.
The RNA polymerase complex of SARS-CoV and SARS-CoV-2 supports the transcription and replication of their approximately 30,000-nucleotide viral RNA genomes. It is the largest and most complex RNA synthesis machinery among RNA viruses. As shown in the illustration to the right, the multi-subunit SARS-CoV polymerase complex is composed of a number of Nsps including viral RdRp (“Nsp12”), processivity factors (“Nsp7”, “Nsp8”), a proofreading exonuclease, a N7-methyl transferase (“Nsp14”), and a helicase (“Nsp13”). The Nsp12 protein contains two domains, a RdRp core, which is the catalytic subunit incorporating ribonucleotides into RNA templates, and an N-terminal NiRAN domain, the function of which was previously unknown.
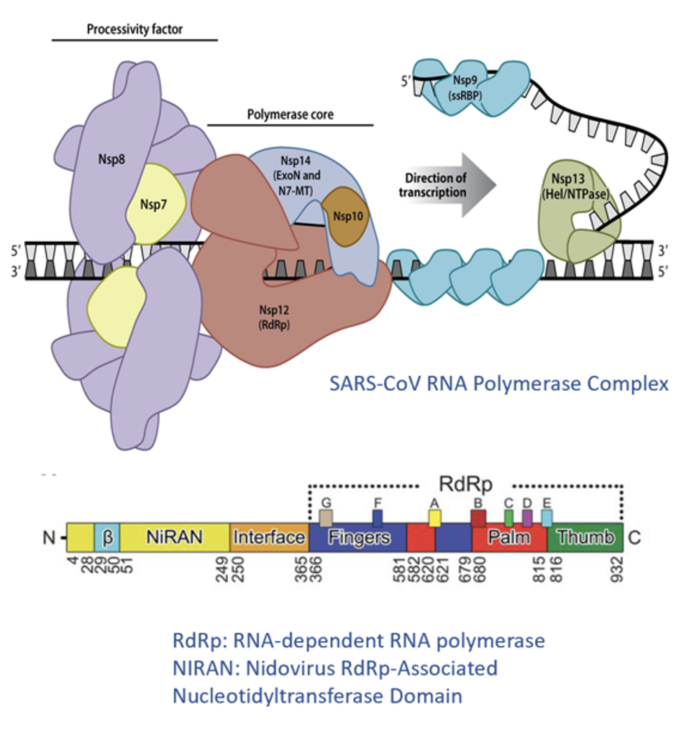
Our collaborators have discovered that SARS-CoV initiates viral RNA synthesis utilizing two distinct pathways: one protein-primed and mediated by the NiRAN through the UMPylation of Nsp8, and the other through de novo synthesis of dinucleotide primers in a NiRAN-independent manner. Importantly, both functions can be inhibited by AT-9010, the active triphosphate metabolite of bemnifosbuvir. Our in vitro studies have shown that AT-9010 binds not only to the NiRAN active site but is also incorporated by the RdRp and functions as a chain terminator. This unique dual mechanism of bemnifosbuvir creates a potentially higher barrier to resistance compared to other direct acting antiviral inhibitors.
Since bemnifosbuvir targets viral RNA polymerase, a highly conserved enzyme critical to viral replication and transcription, it is anticipated that bemnifosbuvir will maintain its antiviral activity even against variants with mutations in the spike (S) protein responsible for the receptor recognition and host cell membrane fusion process. Current COVID-19 variants have lessened the effectiveness of vaccines and eliminated the effectiveness of monoclonal antibodies due to the mutations in the viral spike protein. It is reasonable to anticipate that future variants may also impact the effectiveness of vaccines and monoclonal antibodies which are developed from currently available technologies.
COVID-19 continues to cause infection and disease due to waning immunity and continued emergence of SARS-CoV-2 variants. The CDC has reported that COVID-19 was the fourth leading cause of death in the US in 2022 after only heart disease, cancer and unintentional injury with the majority of COVID-19 deaths occurring in patients aged 65 and older. Older adults and individuals who have risk factors are at a higher risk for developing more serious complications from COVID-19 leading to hospitalization and death.
Infection with SARS-CoV-2 may be asymptomatic or it may cause a wide spectrum of illness ranging from a mild upper respiratory tract infection to severe life-threatening sepsis and multiorgan failure. Commonly reported symptoms include fever, cough, shortness of breath, loss of taste or smell, sore throat, fatigue, headaches, muscle aches, and gastrointestinal disturbance. Symptoms can last two to three weeks, but many patients continue to experience symptoms for many weeks or develop new symptoms, which is now recognized as the post-acute COVID-19 syndrome, or Long COVID. COVID-19 affects people of all ages; however, people who are immunocompromised, elderly, or have certain underlying medical conditions (e.g., chronic heart, lung, and kidney disease; diabetes, obesity, and cancer) are at increased risk of poor outcomes.
COVID-19 is expected to remain a serious endemic threat for an indefinite future period. There are many reasons contributing to the likelihood of COVID-19 remaining an endemic including viral transmission before symptom onset; uneven global rollout of vaccinations; ongoing vaccine hesitancy; limited duration of immunity conferred by both natural infection and vaccination; limited vaccine efficacy against certain SARS-CoV-2 variants; limitations of current oral antivirals such as drug-drug interactions, safety concerns and tolerability; uncertain impact of vaccines on transmission; continuing evolution of the virus evading endogenous and vaccine-induced immunity; and diminution of virus transmission mitigation behaviors, such as wearing masks and social distancing.
At the outset of the COVID-19 pandemic, unprecedented progress was made with both vaccines and treatment options for this novel disease. Despite this progress, there are substantial limitations to currently available vaccines and therapies, including waning immunity to both naturally acquired and vaccine generated immunity, failure of certain populations to mount an adequate immune response to vaccines and lack of efficacy of currently available monoclonal antibodies to currently circulating SARS-CoV-2 subvariants (which have increased transmissibility and the ability to evade neutralizing antibodies). Limitations of current oral antivirals include drug-drug interactions with commonly prescribed medications such as seizure medications, anti-psychotics, anti-coagulants and more, and safety concerns.
As a result, there remains a continued urgent need to develop novel, safe, efficacious, convenient, oral, therapies with low risk of drug-drug interaction for the treatment of COVID-19 that can be utilized as monotherapy and potentially as part of a combination therapy.

SARS-CoV-2 has proven to be endemic and will continue to be prevalent in society. Although vaccines play an important role in improving a patient’s chance of experiencing a milder form of COVID-19, the continuing ability of SARS-CoV-2 to evade vaccines and monoclonal antibodies combined with the limitations of current antivirals, highlight an urgent need for new, oral antiviral treatment options with improved profiles.
Oral therapies protecting against the development of severe infection and transmission remain urgently needed particularly for vulnerable patients who currently have limited treatment options. This includes patients who are unvaccinated, patients who fail to respond to available vaccines, vaccinated patients with waning efficacy, which can occur between three to six months after immunization and patients for whom vaccines and existing treatments are contraindicated due to the potential for drug-drug interactions with commonly prescribed medications.